Ventricular arrhythmias in arrhythmic mitral valve syndrome
Arrhythmic mitral valve syndrome is linked to life-threatening ventricular arrhythmias. The incidence, morphology and methods for risk stratification are not well known. This prospective study aimed to describe the incidence and the morphology of ventricular arrhythmia and propose risk stratification in patients with arrhythmic mitral valve syndrome.
Clinical relevance
- Patients with arrhythmic mitral valve syndrome are at risk of severe arrhythmic events, and risk stratification could be used to guide proper treatment.
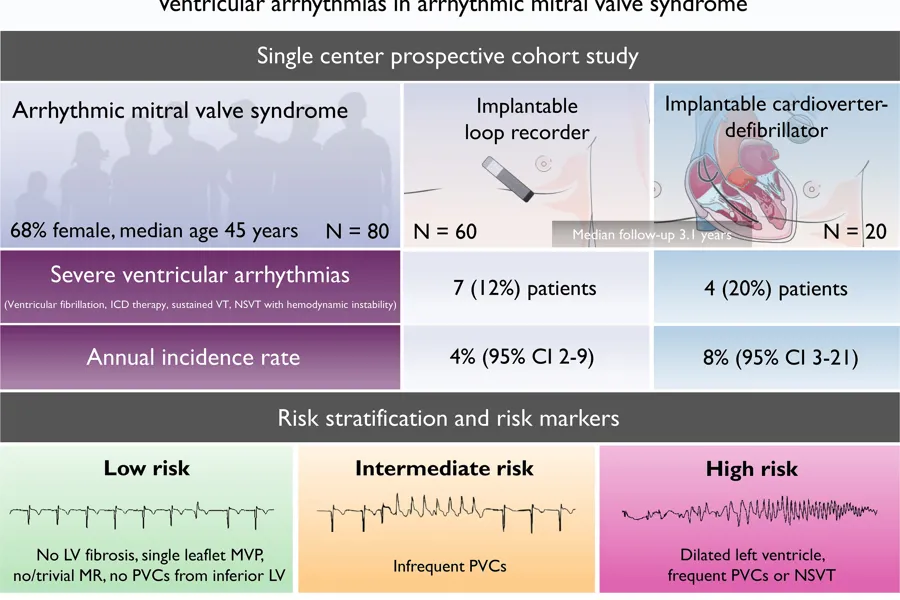
- Patients without prior severe arrhythmic events had a 4% annual incidence of severe ventricular arrhythmic event.
- The annual incidence of severe arrhythmic re-events was 8% among patients with secondary preventive implantable cardioverter-defibrillator.
The yearly incidence of ventricular arrhythmia was high in arrhythmic mitral valve syndrome patients without previous severe arrhythmias using continuous heart rhythm monitoring. The incidence was even higher in patients with secondary preventive ICD. Frequent premature ventricular complexes, non-sustained ventricular tachycardias, greater left ventricular diameter and greater posterolateral mitral annular disjunction distance were predictors of first severe arrhythmic event.
Arrhythmic symptoms are common in these patients, and they frequently have multifocal ventricular arrhythmias arising from the outflow tracts, mitral annulus or left ventricular papillary muscles.
However, current risk markers associated with life-threatening arrhythmias derive from cross-sectional and retrospective studies and lack prospective validation. The incidence of severe ventricular arrhythmias is unknown and clinical decisions on primary prevention implantable cardioverter defibrillator (ICD) remain challenging.
We aimed to describe the incidence of ventricular arrhythmias in patients with arrhythmic mitral valve syndrome. For this, we extended our previous study (Original Investigation; The Mitral Annulus Disjunction Arrhythmic Syndrome, DOI: 10.1016/j.jacc.2018.07.070) by using continuous heart rhythm monitoring. We aimed to provide incidence rates and the morphology of ventricular arrhythmias in patients with and without prior severe ventricular arrhythmia. Additionally, we aimed to explore tools for risk stratification, and strategies for follow-up.
The eligibility criteria used in our study were not meant for clinically selecting patients for ILR monitoring, and should not be used as such in the lack of validation. Additionally, our study was not designed to evaluate the clinical value of systematic ILR implantation in patients with arrhythmic mitral valve syndrome, nor the effect of primary preventive ICD implantation. Whether long-term cardiac monitoring leads to improved prognosis in these patients is still unknown.
The assessment of MVP in presence of MAD is not clearly defined. Future guidelines should address this challenge. We did not assess leaflet redundancy nor curling, which have been associated with ventricular arrhythmias in previous studies
Ventricular arrhythmias in arrhythmic mitral valve syndrome—a prospective continuous long-term cardiac monitoring study | EP Europace | Oxford Academic (oup.com)
Europace. 2022 Oct 18;euac182.
Eivind W Aabel, Monica Chivulescu, Øyvind H Lie, Einar Hopp, Erik Gjertsen, Margareth Ribe, Thomas M Helle-Valle, Thor Edvardsen, Finn Hegbom, Lars A Dejgaard, Kristina H Haugaa
PMID: 36256597
DOI: 10.1093/europace/euac182
Shared under Creative Commons (CC BY-NC 4.0) license.
Relevant clinical trials
Prosjekt #2549105 - Flecainide in Arrhythmic Mitral Valve Prolapse (FLECAPRO) - Cristin
The main goal of this study is to evaluate the effect and safety of adding flecainide to standard beta-blocker therapy to reduce the burden of ventricular arrhythmias in patients with arrhythmic mitral valve prolapse.